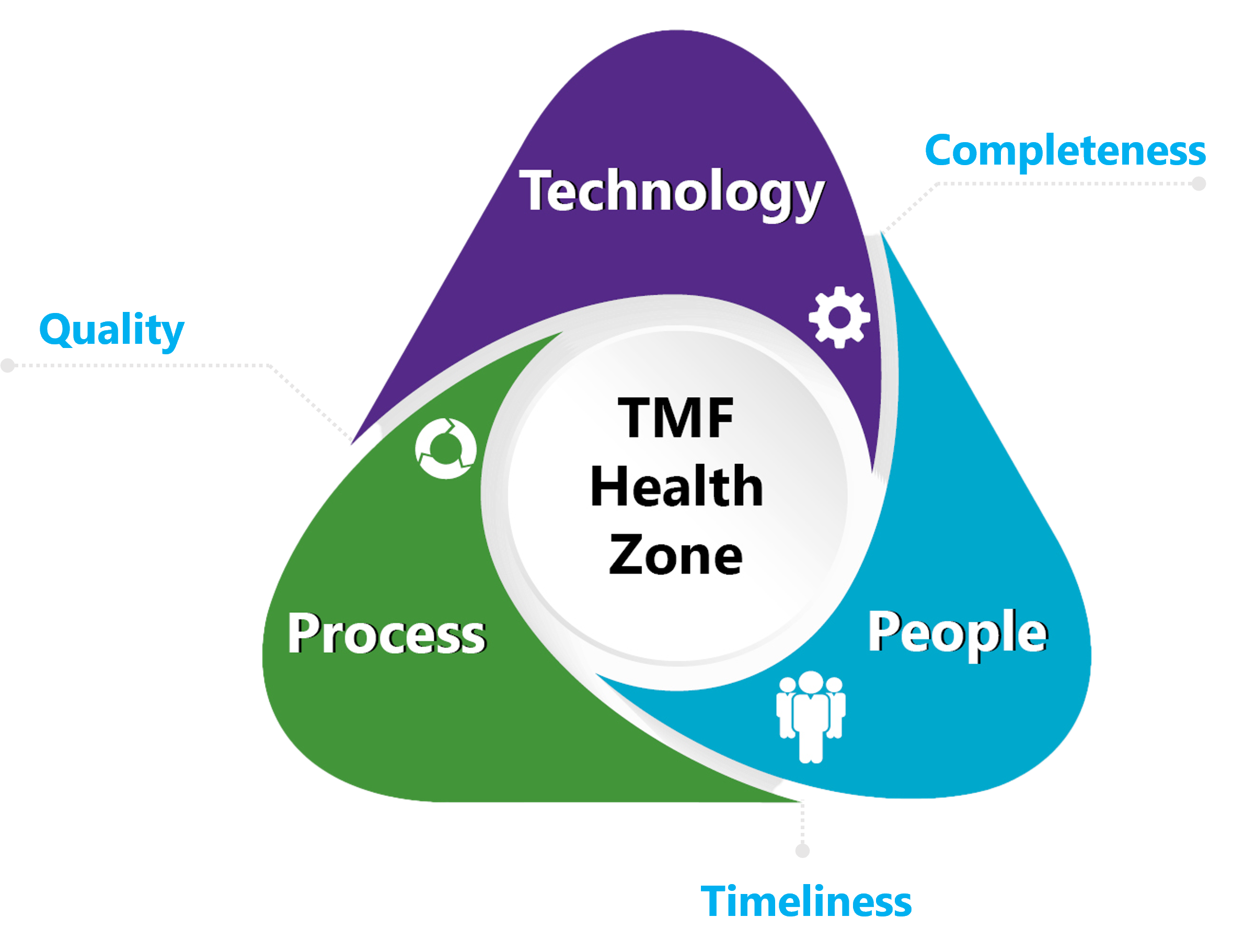
HOW TO SURVIVE A TMF INSPECTION WHEN YOU'RE NOT PREPARED
You’ve just received an inspection notice, and the adrenaline is starting to kick in. When was the last completeness report? Quality review? Did the team finish the latest training on SOPs and the new eTMF system? Where to start?
At a Glance
Phlexglobal’s Expertise Makes a Difference
Innovative Solutions
Reduces Inspection Risk
Offers Global Resources
Increase Output Speed
Brings Quality & Value
Better Value for Money
Phlexglobal Provides Unmatched TMF and Document Management Expert Services

TMF Quality Review
A thorough quality assessment of your TMF, giving you insight into its quality and completeness
TMF Heatmaps
Easily identify potential TMF problem areas and enable a risk-based remediation approach
Migrations & Imports
Help ensure you are meeting GCP standards and regulatory requirements for essential documents
Mergers & Aquisitions
Get a true picture of your acquired drug’s readiness for inspection or submission – and fix any problems


TMF Study Owners
Our TMF practitioners have successfully mastered the challenges you and your teams face every day
And Much More
Continue here to find out how to stay in the TMF Healthzone with expert, scalable services from the global TMF authority
Reduce TMF Misfiles with Next-Generation AI
Latest Insights
Check out our blogs, webinars, articles and more. Connect with our experts and stay up-to-date on all of the trends in the pharmaceutical, biotech and medical device industries.
A Blueprint for Genuine Partnership in TMF Improvement
The Trial Master File (TMF) has evolved from a repository of documents at the end of a study to an integral cog in the regulatory journey. This has led some companies to shift their perspective from

Rewind the clock and take TMF back to basics
Rewind the clock and take TMF back to basics: The paradigm of maintaining simplicity while embracing technology. In 20 years, the Trial Master File (TMF) has moved from paper in dusty lever arch

How to Reduce Risk and Effort When Migrating a Trial Master File
Migrating Trial Master File (TMF) data is a fairly common occurrence, usually driven by one or more of the following scenarios: Implementing a new electronic Trial Master File (eTMF) system
.png)